Michael Faraday
chemistry
|
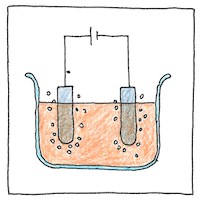
Faraday efficiency
Ideally, during electrolysis, each electron from a battery moves one atom from a compound to the cathode or anode so you could measure the amount of electricity used by weighing the separated portions; however, chemistry can produce losses as separated elements can combine before they get to their electrodes. These losses produce heat instead of producing separated elements.
A charge of one faraday
We used to measure electric charge in faradays. One faraday (the value of the Faraday constant) is the elementary charge times the Avogadro constant, that is, the amount of charge in a mole of electrons.
Waste
A gold miner doesn’t try to sell his empty veins, his crunched quartz, his heaps of worthless slag. When all is said and done, the wasted time and energy won’t matter in the least. Or will it? Wouldn’t it show cause? Human effort? A struggle to do better? Don’t we learn anything from failures?


It is said that Louis Zukofsky burned his juvenilia. He refused to publish poems that he wrote during his years at Columbia. Would they have been a distraction, or would they have provided context for his achievements later?
On the other hand, the rush to publish by Martin Fleischmann and Stanley Pons in 1989 wasted the time and efforts of many people.
See also in The book of science:
Readings in wikipedia: