Rudolf Mössbauer
physics

|
Mössbauer effect
Gases absorb and emit X-rays, but not gamma rays. When gamma rays hit atoms of a gas, the atoms recoil and the energy is lost, so nuclear resonance can not be observed. Rudolf Mössbauer found that solid iridium behaved differently. Nuclear resonance can be observed as solids absorb most of the gamma rays without recoil, and fluoresce, nuclei delivering the energy of impacts to their surrounding crystal lattice.
Mössbauer spectroscopy
Mössbauer spectroscopy observes nuclear interactions, which depend on small shifts of energy levels between particles bound into atomic nuclei. An isomeric shift occurs in an atomic spectra when one nuclear isomer replaces another. A chemical shift is a shift in resonance energy of a nucleus when electrons shift in their orbitals. A quadrupole split occurs in the Mössbauer spectrum when the spin quantum number is greater than one half. A hyperfine-structure split occurs in the Mössbauer spectrum when nuclear multipole moments intereact with internally generated fields. A Zeeman split occurs in an atomic spectrum when exposed to a magnetic field. Mössbauer spectroscopy helps determine relative concentrations of chemicals in a solid and can help show the crystalline size and grain structure of a material.
Non-invasive diagnosis
Geologists and planetary landers on Earth, moon, and Mars probe meteors and other rocks Oil companies set explosives and analyze seismic waves to find oil and gas reservoirs. Sonograms of expectant mothers determines gender. Echocardiograms determine heart irregularities. Home monitors show blood pressure. My doctor taps my back while listening with a stethoscope to evaluate lung functionality.
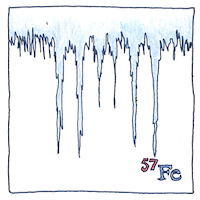


Mössbauer spectroscopy is more penetrating and more precise than Superman’s X-ray vision. Mars landers have used it to discover chemical composition and crystal structure of rocks.
See also in The book of science:
Readings in wikipedia: